B30-24 Amino Acid Composition Analysis in Alternative Protein Food Products
Experimental Details
Sampling
The Biochrom system can provide accurate results from samples in the milligram range or below. However, when studying natural materials, it is preferable to use larger quantities to overcome inhomogeneities in the sample matrix. Material availability is rarely an issue with food products, as was the case here. Therefore, we took 1 g [~1%] samples from each item for this study.
Analysis Options
In some cases, there is interest in quantifying free amino acids in food to identify additives such as flavor enhancers (e.g., monosodium glutamate and aspartame). This is performed via extraction with dilute hydrochloric acid. For meat and meat substitutes, the predominant source of amino acids is protein, making the extraction test inapplicable in this case.
Proteins can be broken down into their constituents by simple hydrolysis with concentrated acid. Unfortunately, this process destroys certain amino acids like cysteine/cystine, methionine, and tryptophan. More comprehensive results are obtained by oxidizing the samples prior to hydrolysis: this converts sulfur-containing amino acids into cysteic acid and methionine sulfone, which are resistant to acid degradation. We applied this method. Tryptophan can be quantified separately via alkaline hydrolysis. We chose a long-established method using barium hydroxide (4).
Procedure
The products were purchased from well-known retailers and manufactured in the UK. Aliquots [1 g] of the meat and plant burgers were transferred to 50 ml round-bottom flasks. 5 ml of oxidation reagent (5) was added to each. The flasks were stored overnight at < 4 °C. After warming to room temperature, sodium metabisulfite [0.84 g] was carefully added to decompose the oxidizing agent.
The flasks were fitted with water condensers and placed in a heating bath. Hydrolysis reagent [12 ml] (6) and anti-bumping granules were added, and the contents were kept under gentle reflux for 24 hours. After cooling, the contents were reduced to a film under vacuum using a rotary evaporator. The residues were taken up in loading buffer (7) and transferred to 50 ml volumetric flasks. 1 ml of internal standard (8) was added, and the volume was made up to the mark with additional buffer.
Chromatography samples [~ 0.5 ml] were filtered and transferred to the autosampler using Uniprep vials (9). A vial containing buffered reference standard with appropriate amino acids and an internal standard of 0.5 mM was prepared (10). A blank vial containing only buffer was also prepared. Chromatography was performed using the "Sodium Oxidised High Performance" program on the Biochrom 30Plus ion-exchange instrument.
For alkaline hydrolysis, 200 mg samples were transferred to 50 ml flasks with 100 mg starch, 6 g barium hydroxide, and 50 ml water. The samples were refluxed overnight. Concentrated hydrochloric acid (~3.5 ml) was added to dissolve the barium hydroxide, and the volume was adjusted to 50 ml.
Note: There were no obvious signs of fat residues in the hydrolyzed samples. In cases where fats are present in large quantities, it is advisable to extract them with a water-immiscible solvent like petroleum ether.
Results
The chromatograms [570 nm channel. Black = standard, red = meat, green = plant-based] showed all amino acids fully resolved within 67 minutes using a standard program. Cysteine and methionine were completely converted to their oxidized forms, indicating sufficient penetration of the reagent. Asparagine was converted to aspartic acid. Hydroxyproline and proline values were taken from the 440 nm channel.
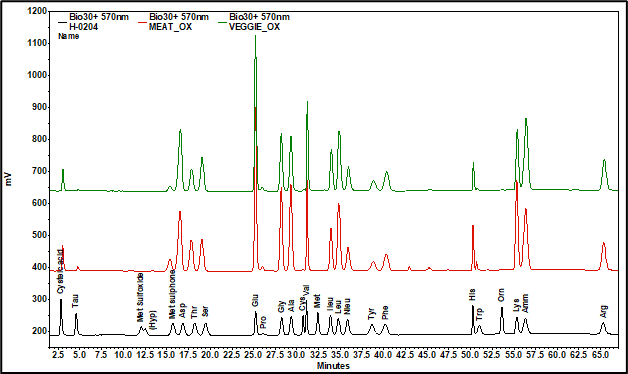
Peak areas were converted into concentrations using individual response factors. Both products were very similar in composition, containing all essential amino acids (Thr, Val, Met, Ile, Leu, Phe, His, Trp, Lys). However, concentrations of Threonine, Histidine, Methionine, and Lysine were significantly higher in the beef product.
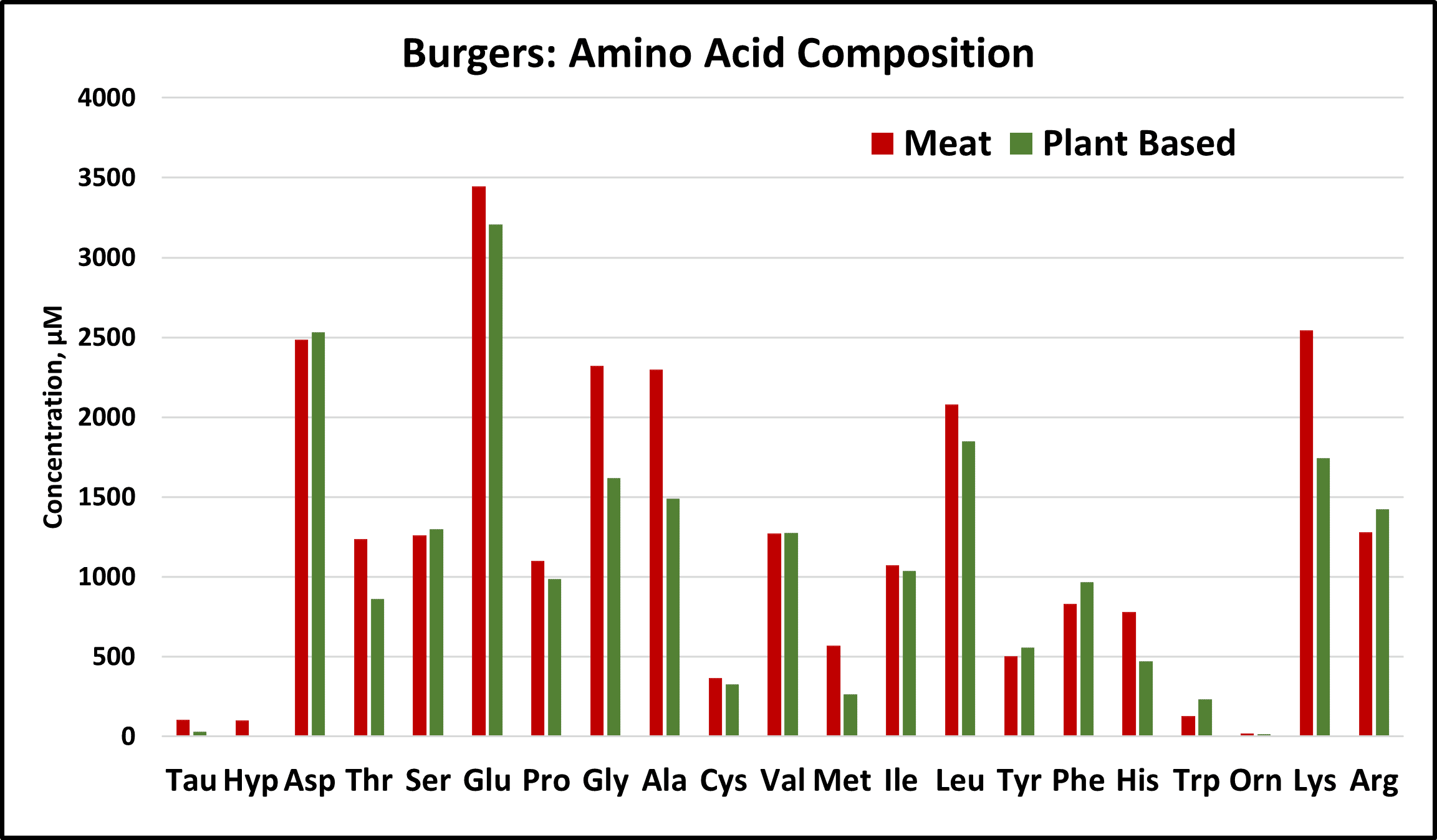
By multiplying the molar concentration of each amino acid by its residual molecular weight and summing them up, the protein composition can be calculated directly. This avoids errors associated with proxy methods like total nitrogen (Kjeldahl). Values of 17% for the meat product and 15% for the plant-based product were determined, largely aligning with the packaging labels.
Conclusion
Amino acid analysis with the Biochrom 30Plus is a simple and highly precise method for separating and quantifying amino acids in a wide range of traditional and alternative protein foods. These results are invaluable for monitoring cell culture compositions, refining flavor profiles, nutritional labeling, and determining protein quality scores like PDCAAS.
Referenzen/Quellen
(1.) Joo ST, Choi JS, Hur SJ, Kim GD, Kim CJ, Lee EY, Bakhsh A, Hwang YH. Eine vergleichende Studie zu den Geschmackseigenschaften von aus Hühner- und Rindermuskeln gewonnenem Fleisch aus Satellitenzellkulturen. Food Sci. Anim. Resour. Januar 2022; 42(1):175-185.
(2.) Amare, E, Mouquet-Rivier, C, Servent, A. , Morel, G. , Adish, A. und Haki, G. Proteinqualität von in Äthiopien angebauten Amaranthkörnern unter dem Einfluss von Poppen und Fermentierung. Food and Nutrition Sciences, 6, 38-48. 2015.
(3.) Mæhre HK, Dalheim L, Edvinsen GK, Elvevoll EO, Jensen I-J. Proteinbestimmung – Die Methode ist entscheidend. Lebensmittel. 2018; 7(1):5.
(4.) A. Dreze und W.S.Reith Biochem. J. 62 (1956) 3P. Auch A. Dreze Bull. Soc. Chim. Biol., 42 (1960) 407
(5.) Bereiten Sie eine Stammlösung aus 45 g Ameisensäure (100 %), 250 mg Phenol und 5 g Wasser vor. Bereiten Sie das Oxidationsreagenz unmittelbar vor Gebrauch zu, indem Sie 18 ml der oben genannten Stammlösung mit 2 ml wässrigem Wasserstoffperoxid (30 %) mischen und 1 Stunde lang bei Raumtemperatur rühren. Vor Gebrauch auf unter 4 °C kühlen.
(6.) Das Hydrolysereagenz wird aus 50 ml konzentrierter Salzsäure und 100 mg Phenol hergestellt, das mit Wasser auf 100 ml aufgefüllt wird.
(7.) Biochrom-Natrium-Ladepuffer #80-2037- 57
(8.) Interne Standard-Stammlösung, 2 mM, hergestellt aus 131 mg Norleucin in 50 ml Ladepuffer.
(9.) Mini-UniPrep-PVDF-Filter, 0,2 µm. Cytiva [Whatman].
(10.) Ein Arbeitsreferenzstandard wurde aus 1 Teil Biochrom Protein Oxidised Standard 2,5 mM, #80-6002-68, 1 Teil Norleucin 2,5 mM und 3 Teilen Ladepuffer hergestellt.
Quelle: Application B30-24 Amino Acid Analysis for Alternative Protein Products (2022) Biochrom Ltd., Cambridge, Großbritannien
Ein Auszug dieses Artikels wurde im Nov. 2025 auch in der Laborfachzeitschrift LABO veröffentlicht, Link zum Beitrag: Aminosäureanalyse: Proteingehalt und Proteinqualität von Fleisch vs. Fleischersatz - LABO

Let’s Discuss Your Specific Requirements
We take the time to truly understand your situation. What types of samples are you analyzing? What is your weekly throughput? What are your specific requirements for reproducibility and system uptime?
In a direct conversation, we’ll determine if the Biochrom B30Plus is the right fit for your workflow - and how we can support you for the long term. No pressure, just practical advice from experts who speak your language.

Proven Performance. For Over 25 Years.

Years on the Market

of All Issues resolved over the phone

Response Time

Key players across 8 countries



